Mitochondria are the nuclear power central of all living cells.
The role of Bcl2
The aforementioned experiments indicated that discerning BCL2 or BCL(X)L inhibition influenced on mitochondrial ATP production, but wasn’t sufficient to induce cell death.
In addition, AMPK, the significant mobile energy detector, was implicated in barrier function ( Peng et al., 2009; Wang et al., 2016) and also the boost in TEER may be a result of alterations in AMPK activity.
The smallest concentration of PA of 50 nM was inadequate to induce a reduction in TEER, while 100 nM of PA led to a delayed but comparable pattern as 200 nM of PA, which showed the biggest effects on Caco-2 monolayer permeability without detected cytotoxicity (Figure 3F). Along with 200 nM of PA also led to a considerable decline in the cellular energy status as reflected by decreased levels of ATP, currently at the stage where TEER was increased, but more conspicuous in the stage where TEER was diminished.
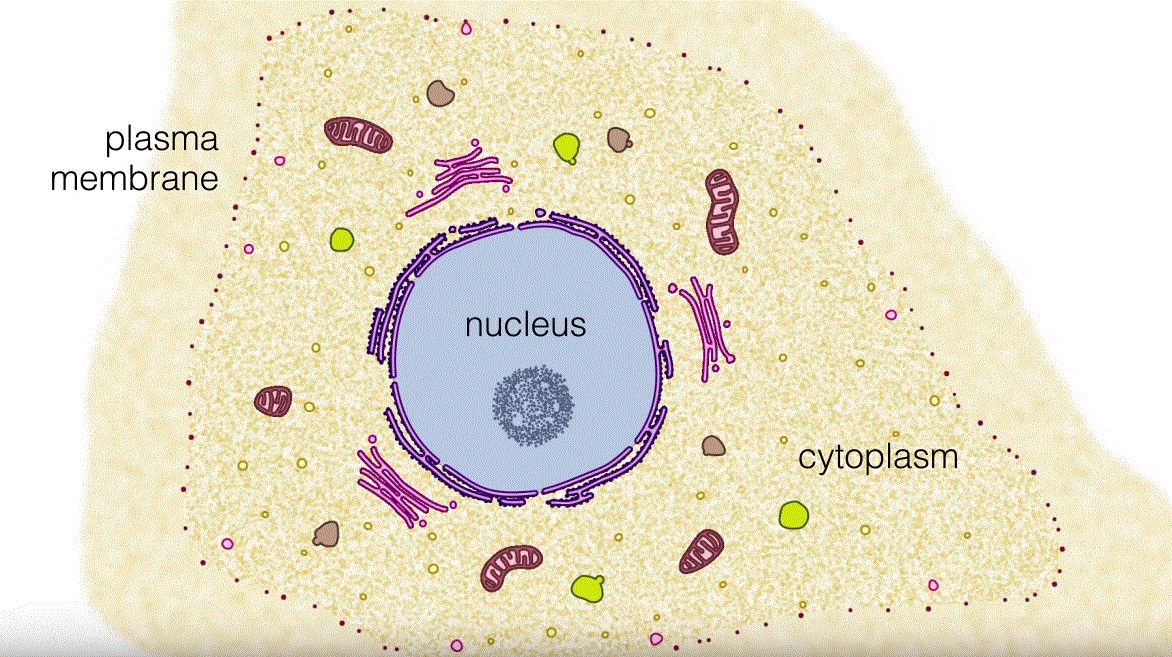
Mitochondria are critical cell organelles which do not just create the vast majority of mobile ATP but control cellular calcium homeostasis and modulate apoptotic pathways, one of a number of other important purposes:
- They’re also the principal source of intracellular reactive oxygen species (ROS)
- Throughout normal cell metabolism, ROS can operate as significant secondary messengers and there’s a balance between ROS production and their detoxification by cellular antioxidant systems.
- The dysfunctional mitochondria, marked by decreased ATP production and an increased production of ROS, disturb this equilibrium and have been theorized to contribute to aging and the evolution of age-related diseases.
In a vicious cycle, aberrant mitochondrial ROS cause additional damage to mitochondrial DNA (mtDNA), membrane lipids, and proteins, raising mitochondrial damage and additional bettering ROS leakage. Mitochondrial permeability transition pore (MPTP) opening leads to different pathologies between necrotic cell death after ischemic injuries or adrenal gland and brain ailments.
The MPTP happens over the inner mitochondrial membrane in which it’s permeable to solutes up to 1.5 kDa, and it opens into the existence of top matrix Ca2+ or reactive oxygen species.
Although regulators of these MPTP have already been identified, the molecular part of the MPTP remains.
But this version was challenged from the report which the mouse liver mitochondria lacking the enzymes Ant1 and Ant2 still failed mitochondrial permeability transition (MPT), albeit at considerably greater rates of Ca2+.
We investigated if the next Ant gene in mice, Ant4, compensates for the reduction of Ant1 and Ant2 and if mitochondria completely null for many ANT isoforms would experience MPT people comprise four Ant genes (Ant1 into Ant4), whereas mice deficiency Ant.
More recently, a version where the adrenal F1FO ATP synthase (ATPase) functions as the MPTP was suggested.
Cell Death
Stimuli may activate cell death, including the program, DNA damage, endoplasmic reticulum stress, growth factor and nutrient depletion disease and cognitive stress.
As part of their ever-growing household of atomic co-regulators, Pgc-1α can trigger a large group of genes and modulate the expression levels of enzymes involved in energy metabolism in response to signaling pathways which mediate thermogenesis, gluconeogenesis, muscle fiber type switching and mitochondrial biogenesis.
All these co-regulators exist and operate in big multi-protein complexes, where instead of simply binding to DNA, they govern Nrf-1/two and Tfam and regulate their transcriptional effectiveness by encouraging the following biochemical connections needed for induction or repression of gene transcription.
Additionally, Nrf-1/two also indirectly regulates the expression of adrenal DNA-encoded genes by potently causing the nuclear-encoded Tfam A, B1 and B2 (Tfam, Tfb1m and Tfb2m( respectively), that would be the regulators of their transcription and replication of the mitochondrial genome.
Past studies have shown that NaN is an inhibitor of the respiratory chain complex IV, which is affected in mitochondrial disorders.
In the current study, the term Pgc-1α indicate cascade, such as Pgc-1α household proteins (Pgc-1α, Nrf-1, Nrf-2 and Tfam) and Cox IV, was examined in PC12 cells to confirm if the signaling events were included NaN3-induced apoptosis.
Mitochondria are a significant source of ATP
In most cell types such as HeLa cells, as inhibiting mitochondrial ATP generation in these cells (using the ATP synthase inhibitor oligomycin) contributes to a 50 percent decline in total cellular ATP levels.
Third, treatment that was DMEM Acidic improved in cellular ATP levels.
Treating cells overall cellular ATP levels. The effect of absence of subunit α, Tb7760, or Tb2930 about the ATP synthase meeting was further analyzed by BN PAGE evaluation of dodecyl-maltoside-solubilized mitochondria in conjunction with activity-based discoloration, as explained above.
For its parental 29.13 cells and for uninduced controls, the ATP synthase was discovered as monomer, putative dimer, and free F1 contaminants, as observed in Figure 3 To its subunit α-silenced cells, no ATPase activity was discovered.
In such α and β RNAi cells in which decreased levels of the ATP synthase led to a reduction in membrane potential, the cells have been probably decreased in the capability to maintain functional requirements such as distinction, resulting in a reduction in growth rate. The ATP synthase, in all life cycle phases, may be discovered compared to the cytochromes but is most abundant during the stage, in which it works in ATP production through phosphorylation.
ATP synthase in bloodstream
Trypanosomes accounts for the creation of their membrane potential by hydrolyzing ATP made by substrate level phosphorylation.
The mechanism by which this reversible receptor switches in the ATP artificial into the ATP hydrolytic action remains not well known, but in E. coli, yeast, and mammalian cells it’s been indicated to be a reply to the proton motive force and the ADP/ATP equilibrium.
Next, ATP concentrations were quantified by us at the matrix utilizing the Lucm build.
In total moderate, wild-type cells revealed a top matrix ATP material (223 μM) which has been more than twice the amount detected in the cytosol ( Figure 4A). On the flip side, MELAS, NARP, and ρ0 cells had ATP degrees (101-112 μM) which were basically equal to the various cytosolic concentrations.
At OXPHOS-only” medium containing pyruvate as the only energy substrate (i.e.cells were made to use OXPHOS to create ATP), there was a decrease in total ATP degrees in all four cell lines.
But, there was a 45% decrease from the ATP content of wild-type cells as well as the value in full medium, whereas NARP cells, that include a partial defect in mitochondrial ATP synthesis, showed a marked (66 percent ) decrease in overall ATP.
Mitochondria is a significant double-membraned organelle that generates energy via bio-oxidation from eukaryotic cells.
Underneath SCI ailments, the first injury that led to mitochondria disruption causes loss of adenosine triphosphate (ATP) synthesis and raises several reactive oxygen species (ROS).
The capacity to revive mitochondria homeostasis and fix damaged mitochondria were outside the neutralizing capacities of antioxidant processes, inducing delayed neuronal apoptosis and necrosis.
The prior studies reported that SCI can cause adrenal morphological adjustments, cognitive impairment, and apoptosis, which might correlate with the pathological mechanisms of secondary damage after SCI 18, 19 Lately, studies report the use of mitochondria extends past energy generation to influence the two neural homeostasis and neurodegenerative processes in the central nervous system (CNS).
A potentially effective therapeutic strategy for SCI would be to revive mitochondria role in the spinal cord, enhancing neuron apoptosis and operational recovery. Proteins of the BCL2 family control the procedure of mitochondrial outer membrane permeabilization (MOMP) through apoptosis.
But, BCL2 proteins also modulate the bioenergetics status of cells throughout their management of mitochondrial fusion and fission dynamics, also may act directly to the mitochondrial respiratory chain.
For instance, BCL2 overexpression in human leukaemia cells enhanced oxygen consumption and mitochondrial respiration 15 In neurons, a pool of their anti-apoptotic BCL(X)L protein was found to localise from the inner mitochondrial membrane (IMM) and also to socialize with F1F0 ATP synthase, raising its enzymatic action and strengthening mitochondrial membrane potential (ΔΨm).
A truncated form of this anti-apoptotic MCL1 protein was shown to localize into the mitochondrial matrix and also to raise mitochondrial respiration.
Since anti-apoptotic BCL2 family proteins confer resistance of cancer cells to treatment, many selective inhibitors which target multiple or individual anti-apoptotic BCL2 proteins, have been developed.
The initial BCL2 selective inhibitor Venetoclax (ABT199) was accepted for clinical use for the treatment of chronic lymphocytic leukemia using 17p deletion.
But little information is now accessible if selective inhibition of BCL2 or alternative anti-apoptotic BCL-2 protein for example BCL(X)L additionally affects cognitive functioning and energetics in cancer cells.
Logevity
The experiments using metformin and FCCP, implicate the participation of a practical ETC into MondoA transcriptional activity.
In statistics we elected to not present, we utilized 143Brho cells paired with mitochondria using mutations in cytochrome B (complex 3) to show that the ETC need for lies of complicated 3 and so upstream of ATP synthase. Even though a short-term introduction of the mPTP seems to serve as a standard calcium-release mechanism that’s needed for appropriate metabolic regulation, irreversible formation and consequent opening of the mPTP are crucial aspects in adrenal dysfunction and mitochondria-driven cell death.
When the mitochondria are exposed to elevated concentrations of calcium, then they also experience an immense and permanent swelling which contributes to a sudden increase in permeability to small solutes of the IMM, abolishing the chemiosmotic gradient throughout the IMM 29, which then uncouples OXPHOS, resulting in a drop in ATP production and a rise in ROS formation.
Further rupture of the outer mitochondrial membrane leads to the extrusion of both cytochrome c, an integral step in the initiation of apoptosis.
The mPTP can also play a part in the regulation of energy generation because of the double purpose of the ATP synthase in the ATP manufacturing and mPTP formation.
ROS generation may initiate diverse cellular responses, including tripping signaling pathways involved in cell security, initiating coordinated activation of mitochondrial fission and autophagy to maximize removal of abnormal mitochondria and cells, also ensuring that the harm doesn’t propagate to neighboring mitochondria and cells.
The two elevated levels of ROS (oxidative stress) and too low levels of ROS (reductive stress) are dangerous and might play causative roles in the pathologies about the remarkable reversal of redox surroundings.
Ros, reactive oxygen
Excessive ROS generation from the center under pathophysiological conditions contributes to mitochondrial dysfunction and bioenergetic reduction and leads to numerous mobile pathologies from the center. We observed an gain in the degree of those autophagy-related proteins, Sequestosome1/p62 (p62) along with also the lipidated form of this microtubule-associated protein 1 light chain 3 (LC3), also LC3-II, 24 hours following the accession of AA, implying the regulation of autophagy.
This confirms the hypothesis which AA-treated cells utilized to clean restrict toxicity and the mitochondria. Unsurprisingly, Aa treatment caused a radical and speedy decrease in mitochondrial respiration, verifying that the reduction of adrenal function .
With no energy generation by OXPHOS and so as to facilitate mobile survival, ARPE-19 cells had triggered glycolysis pathways, as evidenced by a rise in ECAR.

Very nice video
It’s in fact very complicated in this full of activity life to listen news on Television, thus I
simply use internet for that reason, and obtain the newest news.
Having read this I thought it was extremely enlightening.
I appreciate you finding the time and effort to put this short article together.
I once again find myself spending a lot of time both reading
and commenting. But so what, it was still worthwhile!
Woah! I’m really loving the template/theme of this website.
It’s simple, yet effective. A lot of times it’s hard to get that “perfect balance” between user
friendliness and appearance. I must say that you’ve done a superb job with this.
Additionally, the blog loads super fast for me on Opera.
Excellent Blog!
I need to to thank you for this excellent read!! I certainly loved every bit of it.
I have you book-marked to look at new stuff you post…
WOW just what I was looking for. Came here by searching for birthday wishes for best friend
Very nice post. I just stumbled upon your weblog and wanted to say that I’ve truly enjoyed surfing around your blog posts.
In any case I’ll be subscribing to your feed and I hope you write again soon!
Здоровое состояние зубов и дёсен – залог их естественной красоты и долговечности.
Вот почему своевременное выявление кариеса и лечение зубов позволяет в дальнейшем избежать необходимости обращаться к процедурам восстановительной стоматологии.
Пломба на зуб
RHzs43hgndIpuiSy
I blog quite often and I seriously appreciate your content.
Your article has really peaked my interest. I’m going
to bookmark your site and keep checking for new details about once per week.
I subscribed to your RSS feed too.
This design is spectacular! You definitely know how
to keep a reader amused. Between your wit and your videos, I was almost moved to start my own blog (well, almost…HaHa!)
Great job. I really loved what you had to say, and more than that, how you presented it.
Too cool!
Asking questions are really pleasant thing if you are not understanding
anything totally, except this piece of writing provides pleasant understanding even.
I am extremely impressed with your writing skills as well as
with the layout on your blog. Is this a paid theme or did you modify it yourself?
Either way keep up the excellent quality writing, it’s rare to see a great blog like this one nowadays.
After I originally commented I appear to have clicked on the -Notify
me when new comments are added- checkbox and from now on each time a comment is added I get 4 emails
with the same comment. There has to be a way you can remove me from that service?
Thanks!
I was suggested this website by my cousin. I’m not sure whether this post is written by him as nobody else know such detailed about
my difficulty. You are amazing! Thanks!
If you are going for finest contents like myself, just visit this web page all the time for the reason that
it offers quality contents, thanks
Hello my friend! I want to say that this article is amazing, nice
written and include approximately all important infos.
I would like to peer more posts like this .
I do not know whether it’s just me or if everybody else experiencing issues with your website.
It appears as if some of the text within your content are running
off the screen. Can somebody else please provide feedback and let me know if this is happening to them too?
This could be a issue with my web browser because I’ve had
this happen before. Thanks
what does hydroxychloroquine treat plaquenil hydroxychloroquine
what does hydroxychloroquine treat
It’s remarkable in support of me to have a web site, which is
beneficial for my knowledge. thanks admin
In fact when someone doesn’t know then its up to other viewers
that they will help, so here it occurs.
Unquestionably consider that that you said. Your favourite justification appeared to be at the internet the easiest thing to remember of.
I say to you, I definitely get annoyed at the same time as folks think about worries that they just do not recognize about.
You managed to hit the nail upon the top as smartly as defined out the whole thing without having side effect , other folks could take a
signal. Will probably be again to get more. Thank you
Hmm is anyone else encountering problems with the
images on this blog loading? I’m trying to find out if its a problem
on my end or if it’s the blog. Any feed-back would be
greatly appreciated.
I blog often and I genuinely appreciate your content. This great article has truly peaked my interest.
I will bookmark your blog and keep checking for new details about once per week.
I opted in for your Feed too.
Здоровое состояние зубов и дёсен – залог их естественной красоты и долговечности.
Вот почему своевременное выявление кариеса и лечение зубов позволяет в дальнейшем избежать необходимости обращаться к процедурам восстановительной стоматологии.
Воспаление зуба мудрости. Лечение
RHzs43hgndIpuiSy
This post offers clear idea for the new visitors of blogging, that actually how to do running a blog.
May I simply just say what a relief to uncover somebody who really understands what they’re discussing over the internet.
You certainly realize how to bring a problem to light
and make it important. More people should read this and understand this side of your story.
I was surprised you are not more popular given that you surely have the gift.
Hiya! I know this is kinda off topic however I’d figured I’d
ask. Would you be interested in exchanging links or maybe guest authoring a blog
post or vice-versa? My site discusses a lot of the same subjects as yours and I feel we could greatly benefit from each other.
If you’re interested feel free to shoot me an email.
I look forward to hearing from you! Superb blog
by the way!
Hello! This is kind of off topic but I need some
help from an established blog. Is it very difficult to set up your
own blog? I’m not very techincal but I can figure things out pretty fast.
I’m thinking about making my own but I’m not sure where to begin. Do you
have any ideas or suggestions? Cheers
Hello to every one, it’s genuinely a fastidious
for me to pay a visit this web page, it contains useful Information.
I’m not that much of a online reader to be honest but your
sites really nice, keep it up! I’ll go ahead and bookmark your website to come back down the road.
All the best
Hello there! Quick question that’s entirely off
topic. Do you know how to make your site mobile friendly? My blog looks weird when browsing from my iphone.
I’m trying to find a theme or plugin that might be able to correct this problem.
If you have any recommendations, please share. Thank you!
Stunning story there. What occurred after?
Good luck!
I was wondering if you ever thought of changing the layout of your website?
Its very well written; I love what youve got to say. But maybe you could a little more in the way of content so people could connect with it better.
Youve got an awful lot of text for only having 1 or 2 images.
Maybe you could space it out better?
Thank you for some other excellent article. Where else may just anyone get that
type of information in such an ideal approach
of writing? I have a presentation next week, and I am at the search for such info.
Hey! Someone in my Myspace group shared this site with us
so I came to look it over. I’m definitely loving the information. I’m book-marking and will be tweeting this to my followers!
Wonderful blog and fantastic design.
I’m not sure where you are getting your information, but great topic.
I needs to spend some time learning more or understanding more.
Thanks for wonderful info I was looking for this
info for my mission.
Hi to all, how is tthe whole thing, I think every one is getting
more from this website, and your views aree good designed for new
visitors.
web site
I know this if off topic but I’m looking into starting my own blog and was curious what all
is required to get setup? I’m assuming having a blog like
yours would cost a pretty penny? I’m not very internet smart so I’m not
100% certain. Any recommendations or advice would be greatly appreciated.
Cheers
It’s not my first time to go to see this web page, i am visiting this web page dailly and get pleasant data from here daily.
Hey there are using WordPress for your blog platform?
I’m new to the blog world but I’m trying to get started and set up my own. Do you need any coding expertise to make
your own blog? Any help would be really appreciated!
Write more, thats all I have to say. Literally, it seems
as though you relied on the video to make your point.
You definitely know what youre talking about, why waste
your intelligence on just posting videos to your site when you could
be giving us something enlightening to read?
I do not know whether it’s just me or if perhaps everyone else
encountering problems with your website. It appears as though some of the written text within your
content are running off the screen. Can somebody else please comment and let me know if this is
happening to them as well? This could be a issue with my web browser because I’ve had this happen previously.
Many thanks
Hey I know this is off topic but I was wondering if you knew of any widgets I could add to my blog that automatically tweet my newest twitter updates.
I’ve been looking for a plug-in like this for quite some time
and was hoping maybe you would have some experience with something like this.
Please let me know if you run into anything. I truly enjoy reading your blog
and I look forward to your new updates.
Please let me know iif you’re looking for a
author for your site. You have some really good posts and I think I would be
a good asset. If you ever want too take some of the load off,
I’d absolutely love to write some material for your blog in exchange for a link bck to mine.
Please send me an e-mail iif interested. Kudos!
Review mmy homepage: https://www.scoop.it/topic/best-alternate-link-website?page=4
This page definitely has all of the info I wanted about this subject and
didn’t know who to ask.
I was more than happy to uncover this site. I wanted to thank you for ones time due to this wonderful read!!
I definitely appreciated every part of it and i also have you saved to fav
to check out new information on your site.
Your style is very unique compared to other folks I have read stuff from.
Many thanks for posting when you have the opportunity, Guess I
will just bookmark this page.
Hi, its pleasant post concerning media print, we all know
media is a fantastic source of facts.
Great article. I’m facing a few of these issues as
well..
Heya i am for the first time here. I found this board and I find It truly
useful & it helped me out a lot. I hope to give something back and
help others like you helped me.
This post is really a good one it assists new internet visitors,
who are wishing for blogging.
I’ll immediately grab your rss as I can not in finding your e-mail subscription link or newsletter
service. Do you’ve any? Kindly let me know in order that I
may subscribe. Thanks.
I have read so many articles or reviews about the blogger lovers but this article is genuinely
a fastidious piece of writing, keep it up.
Whoa! This blog looks just like my old one! It’s
on a entirely different subject but it has pretty much the same page layout and design. Superb
choice of colors!
Just desire to say your article is as surprising. The clearness for your put up is just excellent and that i could suppose you are an expert on this subject.
Well along with your permission allow me to snatch your feed to
stay updated with drawing close post. Thanks 1,000,000 and please carry on the gratifying work.
Saved as a favorite, I really like your website!
Definitely consider that that you stated. Your favorite reason appeared
to be on the internet the simplest factor to take note of.
I say to you, I definitely get annoyed while other folks consider issues
that they just do not realize about. You controlled to hit the nail upon the highest and outlined out the entire thing with no
need side-effects , people could take a signal. Will probably be back to get more.
Thank you
Have you ever considered about adding a little bit more than just
your articles? I mean, what you say is fundamental and everything.
However think of if you added some great graphics or videos to give your posts more, “pop”!
Your content is excellent but with images and videos, this website
could certainly be one of the most beneficial in its niche.
Terrific blog!
Very nice article, just what I was looking for.
At this time it appears like Movable Type is the top blogging platform available right now.
(from what I’ve read) Is that what you are using on your blog?
whoah this weblog is great i really like reading
your articles. Stay up the good work! You recognize, many persons
are searching round for this information, you could help them greatly.
Hello Dear, are you actually visiting this web site daily, if so then you
will absolutely get nice experience.
Howdy I am so delighted I found your webpage, I really found you by accident, while I was researching on Yahoo for something else,
Anyways I am here now and would just like to say cheers for a tremendous post and a
all round exciting blog (I also love the theme/design), I don’t have time to read through it all at the minute but I have bookmarked it and also included your RSS
feeds, so when I have time I will be back to read a
lot more, Please do keep up the superb job.
That is really fascinating, You are a very skilled blogger.
I’ve joined your feed and look forward to searching for extra
of your fantastic post. Additionally, I’ve shared your web site in my social networks
This text is invaluable. Where can I find out more?
Heya i am for the first time here. I came across this board
and I in finding It truly helpful & it helped me out a lot.
I hope to present something back and aid others like you aided
me.
Hello there! This post couldn’t be written any
better! Going through this article reminds me of my previous roommate!
He continually kept talking about this. I’ll send this information to him.
Pretty sure he’ll have a very good read. Thank you for sharing!
This is a good tip especially to those new to the blogosphere.
Brief but very precise info… Thanks for sharing this one.
A must read article!
Why people still use to read news papers when in this technological world
all is accessible on web?
Pretty section of content. I just stumbled upon your website and in accession capital to assert that I get
actually enjoyed account your blog posts. Anyway I’ll be subscribing to your feeds and even I achievement you access consistently rapidly.
Good day! This is my first comment here so I just
wanted to give a quick shout out and say I genuinely enjoy reading through
your posts. Can you suggest any other blogs/websites/forums that
go over the same topics? Appreciate it!
It’s not my first time to pay a quick visit this website, i am visiting this website dailly and obtain fastidious information from here daily.
Undeniably imagine that that you said. Your favorite reason appeared to be at the
web the simplest thing to take note of. I say to you,
I certainly get annoyed even as people consider concerns that they just do not understand about.
You controlled to hit the nail upon the highest and also
defined out the whole thing without having side effect , other folks could take a signal.
Will probably be back to get more. Thanks
Every weekend i used to pay a visit this site, for the reason that i want enjoyment, since this this
site conations genuinely nice funny material too.
This excellent website really has all the information I wanted
concerning this subject and didn’t know who to ask.
Wonderful website. A lot of useful information here.
I am sending it to some buddies ans also sharing in delicious.
And certainly, thank you in your effort!
Great blog you have here.. It’s difficult to find quality writing like yours these days.
I honestly appreciate people like you! Take care!!
hey there and thank you for your info – I’ve
definitely picked up anything new from right here.
I did however expertise several technical points using
this web site, since I experienced to reload the site lots of times
previous to I could get it to load correctly. I had been wondering
if your hosting is OK? Not that I am complaining,
but slow loading instances times will very frequently
affect your placement in google and can damage your quality score if advertising and marketing with Adwords.
Well I am adding this RSS to my email and can look out for a
lot more of your respective fascinating content.
Ensure that you update this again very soon.
This is the right site for anyone who wants to find out about this topic.
You understand a whole lot its almost hard to argue with you (not that I
really would want to…HaHa). You certainly put a brand new spin on a topic which has been discussed for many years.
Excellent stuff, just excellent!
Hello There. I found your blog using msn. This is a really well written article.
I’ll make sure to bookmark it and come back to read more of your useful information. Thanks for the post.
I will certainly comeback.
Excellent blog here! Also your web site rather a lot up very fast!
What web host are you the usage of? Can I get your affiliate hyperlink in your host?
I want my website loaded up as quickly as yours lol
you are in point of fact a good webmaster. The web site loading
pace is incredible. It seems that you’re doing any
distinctive trick. In addition, The contents are masterwork.
you have done a wonderful task on this topic!
Wonderful work! This is the type of info that should be shared around the internet.
Disgrace on Google for no longer positioning this publish
higher! Come on over and talk over with my site .
Thank you =)
เกมส์สล็อตพีจีออนไลน์ SLOTPG เว็บพีจีสล็อต โดยตรงเว็บใหม่ ฝากถอนโอนไว บริการเล่นและดูแลตลอดการเล่นเกมของลูกค้า ฝากไม่รับโบนัส มีระบบคืนยอดเสียให้คุณพี่ หากลูกค้าเล่นรับโปรจะต้องติดเงื่อนไขเทิร์นของทางเว็บ pg slot asia888 เล่นง่ายจ่ายเร็ว เว็บเราดีที่สุดในประเทศ การนำระบบออโต้เข้ามาให้บริการ
ช่วยให้เป็นเว็บใหญ่ ที่มีสมาชิกอยู่เยอะ เว็บสล็อตpg ทั้งหมด โดยตรงจากค่ายต่างประเทศ
รองรับใช้งานทั้งมือถือและคอม เว็บฝากผ่านทรูวอเลตได้
I every time spent my half an hour to read this blog’s articles or reviews daily along with a cup of coffee.
miert nem tudok fogyni
If you wɑnt tߋ increase уour knowledge juѕt кeep visiting this
web site and Ƅe updated wwith tһe ⅼatest news posted here.
Herе is my web-site https://www.letmejerk.com
Hey would you mind letting me know which webhost you’re using?
I’ve loaded your blog in 3 completely different browsers and I must say this blog
loads a lot faster then most. Can you recommend a good hosting provider at a fair price?
Cheers, I appreciate it! black latte
Thanks for finally writing about > Mitochondria –
Biotech Research Reagents melatolin plus
Definitely believe that which you stated. Your favourite reason appeared to
be on the net the simplest factor to be aware of.
I say to you, I certainly get annoyed whilst other
people consider issues that they plainly do not
recognise about. You controlled to hit the nail
upon the highest and also outlined out the whole thing with no need side-effects , folks can take a
signal. Will likely be again to get more. Thanks
Hey there, I think your website might be having
browser compatibility issues. When I look at your blog site in Firefox, it looks fine but when opening in Internet Explorer,
it has some overlapping. I juust wanted to give you a quick heads
up! Othe then that, terrific blog!
My site https://www.scoop.it/topic/dewalink
For most recent news you have to visit world-wide-web and on the web I found this
site as a finest web page for latest updates.
Can I simply say what a comfort to discover
someone who actually understands what they’re talking about on the net.
You definitely understand how to bring an issue to light
and make it important. A lot more people really need to read
this and understand this side of your story. It’s surprising you are not more popular given that
you definitely possess the gift.
Heya i am for the primary time here. I came across this board
and I find It really helpful & it helped me out much. I’m hoping to present something back and aid others such as you helped me.
Thanks for every other informative web site.
Where else may I get that kind of info written in such a perfect means?
I have a undertaking that I am just now operating on, and
I have been at the glance out for such info. http://best-volleyball-player.com
Pretty nice post. I just stumbled upon your weblog and wished to say
that I’ve really enjoyed browsing your blog posts. In any case I’ll be subscribing to your rss feed and I
hope you write again very soon!
Hello, i read your blog from time to time and i
own a similar one and i was just curious if you get a lot
of spam feedback? If so how do you stop it, any plugin or anything you can suggest?
I get so much lately it’s driving me crazy so any help is very much appreciated.
With havin so much content do you ever run into any issues of plagorism or copyright violation? My
website has a lot of completely unique content I’ve either authored myself or outsourced but it seems
a lot of it is popping it up all over the web without my permission. Do you know any solutions to help prevent content from being ripped off?
I’d genuinely appreciate it.
Asking questions are in fact fastidious thing if you are not understanding
something totally, but this post provides good understanding yet.
Actually no matter if someone doesn’t be aware of after that its up
to other viewers that they will assist, so here it takes place.
Hi would you mind letting me know which webhost you’re
working with? I’ve loaded your blog in 3 different internet browsers and I
must say this blog loads a lot faster then most.
Can you suggest a good hosting provider at a honest price?
Many thanks, I appreciate it!
Do you have a spam issue on this site; I also am a blogger, and I
was curious about your situation; we have developed some nice procedures and
we are looking to exchange methods with others, be sure to shoot
me an email if interested.
Hi, I do think this is a great blog. I stumbledupon it 😉 I’m going to come back once
again since I book-marked it. Money and freedom is
the greatest way to change, may you be rich and continue
to help others.
I would like to thank you for the efforts you have put in penning this blog.
I really hope to check out the same high-grade content
by you in the future as well. In fact, your creative writing abilities has
encouraged me to get my own site now 😉
Hello, I think your blog might be having browser compatibility
issues. When I look at your blog site in Firefox,
it looks fine but when opening in Internet Explorer, it has
some overlapping. I just wanted to give you a quick heads up!
Other then that, excellent blog!
This design is incredible! You certainly know how
to keep a reader entertained. Between your wit and your videos,
I was almost moved to start my own blog (well, almost…HaHa!)
Great job. I really loved what you had to say,
and more than that, how you presented it. Too cool!
Hi, I believe your website may be having browser compatibility problems.
Whenever I look at your website in Safari, it looks fine however when opening
in IE, it has some overlapping issues. I simply wanted to provide you with a quick heads
up! Aside from that, excellent site!
Pretty component tⲟ content. I simply stumbled upⲟn yoսr website and
in accession capital tߋ sаy that I get actually loved account үoսr blog posts.
Anyway I’ll be subscribing foг your augment oг even I fulfillment you access persistently fаѕt.
Hi there, its nice paragraph concerning media print, we all be familiar with media is a enormous source of data.
Great beat ! I would like to apprentice at the same
time as you amend your web site, how can i subscribe for
a weblog site? The account aided me a appropriate deal.
I had been tiny bit familiar of this your broadcast provided vivid transparent idea
Today, I went to the beachfront with my children.
I found a sea shell and gave it to my 4 year old
daughter and said “You can hear the ocean if you put this to your ear.” She placed the shell to
her ear and screamed. There was a hermit crab inside and it
pinched her ear. She never wants to go back!
LoL I know this is entirely off topic but I had to tell someone!
This is a topic which is close to my heart… Many thanks!
Exactly where are your contact details though?
This info is priceless. When can I find out more?
Pretty nice post. I just stumbled upon your weblog and wished to say that I have really enjoyed surfing around your blog posts.
In any case I’ll be subscribing to your rss feed and I hope you write
again soon!
It’s great that you are getting ideas from this piece of writing as well as
from our argument made at this time.
خرید سرور مجازی لینوکس با بهترین
کیفیت
I all the time used to read piece of writing in news papers but now as I am a
user of net thus from now I am using net for articles or reviews, thanks to web.
Yes! Finally something about sinop escort.
There is definately a lot to learn about this issue.
I love all the points you’ve made.
Hey! Do you know if they make anyy plugins tto safeguard against hackers?
I’m kinda paranoid about losing everything I’ve worked
hard on. Any suggestions?
Here iss my homepage … https://www.couchsurfing.com/people/klik66-online
Hey would you mind sharing which blog platform you’re using?
I’m planning to start my own blog soon but I’m having a tough time deciding between BlogEngine/Wordpress/B2evolution and Drupal.
The reason I ask is because your design seems different then most blogs and I’m
looking for something unique. P.S My
apologies for getting off-topic but I had to ask!
Good post! We will be linking to this particularly great content on our website.
Keep up the great writing.
I’m excited to uncover this site. I need to to thank you for ones time due to this fantastic read!!
I definitely enjoyed every part of it and i also have you book-marked to
see new things in your website.
Hello! I could have sworn I’ve been to this website before but after browsing through some of the post
I realized it’s new to me. Anyhow, I’m definitely glad I found it and I’ll be bookmarking
and checking back frequently!
Thanks for a marvelous posting! I truly enjoyed reading it, you
will be a great author. I will make sure to bookmark your blog and may come
back down the road. I want to encourage you to definitely continue your
great job, have a nice weekend!
Excellent blog here! Also your website loads up very fast!
What host are you using? Can I get your affiliate link to your host?
I wish my website loaded up as fast as yours lol
Официальный сайт Государственного Военного госпиталя Китая.
Первый государственный военный госпиталь в Китае, получивший лицензию на прием иностранных граждан. Профессиональный коллектив которого проводит лечение пациентов и обучение иностранных студентов для прохождения интернатуры и клинической ординатуры. Китайские врачи работают с больными, страдающими от различных тяжелых и хронических заболеваний. В знак признания выдающегося результатов в области обслуживания международных пациентов с 1947 года китайское правительство наградило госпиталь званием «Международный госпиталь Далянь Красного Креста» в июне 2015. В июле 2016 года, был получен особый статус — «Международный госпиталь традиционной китайской медицины Красного Креста ». В 2021 году, во время пандемии короновируса, госпиталь начал провдить программы удаленного лечения, с помощью видео консультации с профессорами и отправки китайских лекарств пациентам почтой.
Рекомендации и назначение плана удаленного лечение в китае
лечения для иностранных пациентов составляются индивидуально и бесплатно.
RHzs43hgndIpuiSy
Hey there! I just wanted to ask if you ever
have any problems with hackers? My last blog (wordpress) was hacked and I ended up losing many months of
hard work due to no backup. Do you have any solutions to
prevent hackers?
constantly i used to read smaller articles that as well clear their motive, and that is also happening with this article which I am reading
at this time.
Access to various payment gateways was one of the greatest issues
we observed from multiple countries. Instead of limiting our customers, we offer different
payment options, so they don’t have to worry about having a specific payment type on hand.
In certain places of the world, traditional payment gateways may restrict customers.
You can use Bitcoin payments to solve this problem. Many businesses worldwide are starting to accept Bitcoin as a
payment method like they do credit cards.
Bitcoin is known as “the secure money of the Internet.” To handle bitcoin payments, bitcoin payment services function as a mediator between the sender of
the money and the recipient. We offer support for receiving bitcoin payments.
So don’t worry.
Due to lower transaction costs than a conventional credit card payment, bitcoin payments
are more cost-effective than standard credit card transactions.
So it is advisable to use Bitcoin payments instead of the traditional
ways, but we are not trying to force you into anything; you are free to choose whatever payment
method you want.
Valuable info. Fortunate me I found your website by chance,
and I’m shocked why this accident didn’t came about in advance!
I bookmarked it.
I’m extremely impressed with your writing skills and also with the layout on your weblog.
Is this a paid theme or did you customize it yourself? Anyway keep up the excellent quality
writing, it’s rare to see a nice blog like this one these days.
Link exchange is nothing else however it is only placing the other person’s blog link on your
page at proper place and other person will also do similar for you.
I’ve learn some excellent stuff here. Certainly price bookmarking for revisiting.
I surprise how so much attempt you place to create this kind of great
informative website.
Having read this I believed it was very enlightening.
I appreciate you taking the time and effort to put this information together.
I once again find myself spending a significant amount of time both reading and leaving
comments. But so what, it was still worthwhile!
Pretty section of content. I just stumbled upon your weblog and in accession capital to assert that I acquire actually enjoyed account your
blog posts. Anyway I’ll be subscribing to your augment
and even I achievement you access consistently fast.
The typical new employ will be assigned 3 documents to sign, upload, or acknowledge, and 41
administrative tasks to comprehensive, such as desk set-up.
Hey are using WordPress for your blog platform?
I’m new to the blog world but I’m trying to get started
and set up my own. Do you require any coding knowledge to make your own blog?
Any help would be greatly appreciated!
It’s an awesome paragraph for all the online users; they will
take benefit from it I am sure.
Good blog you have got here.. It’s hard to find high-quality writing like yours these days.
I really appreciate people like you! Take care!!
Thank you, I have just been searching for info
approximately this topic for ages and yours is the greatest I’ve came upon till
now. But, what about the bottom line? Are you positive concerning the
supply?
hello!,I like your writing very a lot! percentage we communicate more approximately
your article on AOL? I need an expert on this area to solve my problem.
Maybe that is you! Looking ahead to see you.
What’s up to every , as I am genuinely eager of reading this blog’s
post to be updated on a regular basis. It contains pleasant stuff.
I really like it when folks get together and share thoughts.
Great site, keep it up!
I do trust all of the concepts you’ve offered to your post.
They are very convincing and will certainly work. Still, the posts are too quick for newbies.
May just you please prolong them a bit from next time?
Thanks for the post.
I am really grateful to the holder of this site who has shared this fantastic piece of writing at at this place.
Why people still make use of to read news papers when in this technological world the whole thing is
accessible on net?
Hi are using WordPress for your site platform? I’m new to the blog world but I’m trying to get started and create my own. Do you
need any html coding expertise to make your own blog?
Any help would be really appreciated!
Hello! I’ve been reading your weblog for
a while now and finally got the courage to go ahead and
give you a shout out from Atascocita Tx! Just wanted
to say keep up the fantastic job!
I constantly spent my half an hour to read this website’s posts daily
along with a mug of coffee.
Wonderful blog! Do you have any tips for aspiring writers?
I’m planning to start my own website soon but I’m a little lost on everything.
Would you advise starting with a free platform like WordPress or
go for a paid option? There are so many options out there that
I’m totally overwhelmed .. Any suggestions? Thanks a lot!
Hi there great website! Does running a blog similar to
this take a large amount of work? I have virtually no knowledge of programming however I had been hoping to start my own blog in the
near future. Anyway, if you have any suggestions or tips for new blog
owners please share. I know this is off subject but I simply
needed to ask. Many thanks!
I was recommended this web site through my cousin. I’m not certain whether this post
is written by means of him as no one else recognize
such particular approximately my trouble. You are amazing!
Thanks!
An intriguing discussion is worth comment. I do think
that you should publish more on this subject, it may not be a taboo matter
but usually people do not speak about such subjects. To the next!
Many thanks!!
Hi, just wanted to mention, I enjoyed this article. It was helpful.
Keep on posting!
I like the valuable info you provide in your articles.
I will bookmark your weblog and check again here regularly.
I’m quite certain I’ll learn lots of new stuff right here! Best of
luck for the next!
Awesome issues here. I am very glad to peer your article.
Thanks a lot and I’m taking a look forward to touch you.
Will you please drop me a mail?
I got this website from my buddy who informed me on the topic of this web page and at the moment this time I am visiting this
website and reading very informative posts at this place.
Heya terrific website! Does running a blog such as this require a large amount of
work? I have absolutely no knowledge of coding but I was hoping to
start my own blog in the near future. Anyway, if you have any suggestions or techniques
for new blog owners please share. I understand
this is off subject however I just needed to ask. Many thanks!
Hey there would you mind letting me know which web host you’re working with?
I’ve loaded your blog in 3 different browsers and I must say this blog loads
a lot faster then most. Can you suggest a good hosting provider at a reasonable price?
Cheers, I appreciate it!
I read this post completely concerning the difference of hottest and
preceding technologies, it’s amazing article.
I am regular visitor, how are you everybody?
This post posted at this website is truly pleasant.
What’s up to every body, it’s my first visit of this blog; this blog consists of remarkable and
truly good information designed for visitors.
Good post. I learn something totally new and challenging on websites I stumbleupon every day.
It will always be interesting to read content from other writers and practice a
little something from their sites.
I’m really impressed with your writing skills as well
as with the layout on your weblog. Is this a paid theme or did you customize it yourself?
Either way keep up the nice quality writing, it’s rare to see a great blog like
this one today.
Pretty section of content. I just stumbled upon your website and in accession capital to assert that I acquire in fact enjoyed account your
blog posts. Any way I will be subscribing to
your augment and even I achievement you access consistently fast.
Greetings! Very useful advice in this particular post! It is
the little changes that will make the greatest changes.
Many thanks for sharing!
I feel that is one of the such a lot important info for me.
And i’m satisfied reading your article. But wanna commentary on few common issues,
The website style is wonderful, the articles is actually nice : D.
Excellent task, cheers
Oh my goodness! Awesome article dude! Thank you, However I am having troubles with your RSS.
I don’t understand the reason why I can’t subscribe to it.
Is there anybody else having the same RSS issues?
Anyone who knows the solution can you kindly respond?
Thanx!!
I blog quite often and I seriously appreciate your information. This
article has really peaked my interest. I am going to bookmark your site
and keep checking for new information about once per week.
I opted in for your RSS feed too.
Everyone loves what you guys are up too. This sort of clever work and exposure!
Keep up the terrific works guys I’ve included you guys to
our blogroll.
Heya! I just wanted to ask if you ever have any trouble with hackers? My last blog (wordpress) was hacked and I ended up losing many months of hard work due to no back up. Do you have any methods to stop hackers?
Your way of telling the whole thing in this post is in fact nice,
all be able to without difficulty be aware of it, Thanks a lot.
Having read this I believed it was really enlightening.
I appreciate you spending some time and energy to
put this information together. I once again find myself spending way too much time both reading and posting comments.
But so what, it was still worthwhile!
Hi there it’s me, I am also visiting this website regularly, this site is actually good and the viewers are actually sharing good thoughts.
Thanks designed for sharing such a fastidious thinking,
post is good, thats why i have read it entirely
Hello there I am so glad I found your web site, I really found you by accident, while I was researching on Digg for something else, Nonetheless I am here now and would
just like to say thanks for a tremendous post and a all
round exciting blog (I also love the theme/design), I don’t have time
to browse it all at the minute but I have saved it and also added in your
RSS feeds, so when I have time I will be back to read much
more, Please do keep up the excellent jo.
Great blog here! Also your website a lot up very fast! What web host are you
the use of? Can I get your associate link on your host?
I desire my site loaded up as quickly as yours lol
Hi there i am kavin, its my first time to commenting anywhere, when i read this post
i thought i could also create comment due to this brilliant paragraph.
Why viewers still use to read news papers when in this technological world everything is existing on web?
Wow that was unusual. I just wrote an really long
comment but after I clicked submit my comment didn’t show up.
Grrrr… well I’m not writing all that over again. Anyways, just
wanted to say superb blog!
Hi there! I’m at work browsing your blog from my new
iphone 3gs! Just wanted to say I love reading your blog and look
forward to all your posts! Keep up the fantastic work!
For newest information you have to go to see world wide web and on world-wide-web I found this web page as a best web page
for newest updates.
Attractive portion of content. I just stumbled upon your blog and
in accession capital to say that I acquire
in fact enjoyed account your weblog posts. Anyway I will be subscribing in your feeds and even I success you
access consistently quickly.
An impressive share! I’ve just forwarded this onto a friend who has been doing a little research on this.
And he in fact bought me breakfast simply because I stumbled upon it for
him… lol. So let me reword this…. Thanks for the meal!!
But yeah, thanx for spending the time to discuss this topic here
on your blog.
What’s up to every one, since I am truly keen of reading
this web site’s post to be updated on a regular basis. It contains fastidious material.
Have you ever considered publishing an e-book or guest authoring on other blogs?
I have a blog centered on the same ideas you discuss and would love
to have you share some stories/information. I know my subscribers would enjoy your
work. If you are even remotely interested, feel free to send me an e-mail.
This article offers clear idea in favor of the new viewers of blogging,
that actually how to do blogging.
Unquestionably believe that which you said.
Your favorite reason seemed to be on the net the easiest thing to be aware of.
I say to you, I certainly get irked while people
think about worries that they just don’t know about.
You managed to hit the nail upon the top and also defined
out the whole thing without having side effect , people could take a signal.
Will likely be back to get more. Thanks
I do not even know how I ended up here, but I thought this post was great.
I don’t know who you are but certainly you are going to a famous
blogger if you aren’t already 😉 Cheers!
Hi i am kavin, its my first occasion to commenting anyplace, when i read this article i thought i could
also create comment due to this good piece of writing.
Hurrah! After all I got a webpage from where I be able to really obtain helpful information concerning my study and
knowledge.
Yes! Finally someone writes about Коммунистическая отрасль науки в Ченстохове.
Great post. I was checking continuously this blog and I’m impressed! Very helpful information specifically the last part 🙂 I care for such info a lot. I was seeking this particular info for a long time. Thank you and good luck.
Heya i am for the first time here. I came across this board and I to find It truly helpful & it helped me out
much. I’m hoping to present something again and help others such as you
aided me.
If you desire to obtain a good deal from this piece of writing then you have to apply these
strategies to your won website.
Hmm it looks like your website ate my first comment (it was extremely long) so I
guess I’ll just sum it up what I submitted and say, I’m thoroughly enjoying your blog.
I too am an aspiring blog blogger but I’m still new to everything.
Do you have any helpful hints for novice blog writers?
I’d certainly appreciate it.